Alzheimer's Disease Treatments: Which is better?
A Balanced Look at Standard of Care vs. Functional Medicine Approaches for Alzheimer's Disease
With Alzheimer's affecting nearly 1 in 9 Americans over 65, the debate between conventional drug treatments and functional medicine approaches has never been more important. Here is what the science actually says — and what it means for you and your loved ones.

If someone you love has been diagnosed with Alzheimer's disease — or if you have been navigating your own cognitive concerns — you have probably already encountered a dizzying array of information. There are FDA-approved medications, experimental infusions, and an ever-growing world of lifestyle protocols, supplements, and "reversal" programs that promise what conventional medicine has not yet delivered. It is completely natural to feel both hopeful and skeptical at the same time. As a physician, my goal here is not to sell you on any single approach. Instead, I want to walk you through what the science actually shows — the good, the bad, and the complicated — so that you can have a genuinely informed conversation with your own medical team.
Alzheimer's disease currently affects approximately 10.8% of Americans over the age of 65, making it one of the most urgent public health challenges of our time. Despite decades of research and an estimated $40 billion spent on drug development, we still do not have a cure. What we do have are two increasingly distinct schools of thought about how to approach this disease — and in 2025, for the first time, we have meaningful scientific data to compare them directly.
Understanding Alzheimer's: Two Very Different Explanations
Before we can evaluate treatments, it helps to understand how each camp explains what is actually happening in the brain — because the treatment philosophy flows directly from the theory of disease.
The Traditional View: Amyloid is the Enemy
For over three decades, mainstream Alzheimer's research has been dominated by the amyloid cascade hypothesis. In plain terms, this theory holds that a protein fragment called amyloid-beta (Aβ) accumulates in the brain, forms toxic plaques, and sets off a chain reaction that destroys neurons. A second protein, called tau, becomes abnormally twisted ("hyperphosphorylated") as a downstream consequence, further disrupting brain cell function. Under this framework, the logical treatment target is clear: get rid of the amyloid.
Neuroinflammation, oxidative stress, and impaired cellular cleanup pathways also play documented roles in the disease, though traditional treatments have rarely targeted all of these simultaneously. Mainstream research increasingly acknowledges this complexity, even if drug development has been slow to catch up.
The Functional Medicine View: Alzheimer's is a Network Problem
Systems biology researchers, led most prominently by Dr. Dale Bredesen, propose a fundamentally different framework. In this model, Alzheimer's disease is not caused by a single villain (amyloid) but is instead the result of a chronic imbalance between the brain's ability to build and maintain synaptic connections versus the forces tearing them down.
Think of it like osteoporosis. Bone loss happens when the cells that break down bone consistently outpace the cells that build it. Similarly, in this model, cognitive decline occurs when "synapse-destroying" signals chronically overwhelm "synapse-building" ones. The amyloid protein, in this framework, is not simply a mistake — it is actually part of the brain's attempt to protect itself against metabolic stress, toxin exposure, chronic infections, and hormonal withdrawal. When those underlying stressors are left unaddressed, the protective response spirals into damage. Removing the amyloid without fixing what caused it in the first place, proponents argue, is like mopping up a flooded floor without turning off the tap.

This distinction in how each field understands the disease is not merely academic — it has profound implications for how treatment is designed and what outcomes are realistically possible. If Alzheimer's is primarily an amyloid problem, then clearing amyloid should be the goal. If it is a complex network failure driven by dozens of intersecting metabolic, hormonal, and environmental factors, then no single drug can ever be sufficient. Understanding which framework best explains the evidence is one of the most important questions in modern medicine — and the answer may not be as simple as either side claims.
What Does Standard of Care Actually Offer?
It is worth being honest about where conventional Alzheimer's treatment stands today, because many patients and families are surprised — and sometimes devastated — to learn its real limitations.
The Medications You Have Probably Heard Of
For over two decades, the standard pharmacological treatment for Alzheimer's has consisted of two classes of drugs: **cholinesterase inhibitors **(donepezil, rivastigmine, and galantamine) and an NMDA receptor antagonist called memantine. These medications were developed to compensate for the neurotransmitter deficits observed in Alzheimer's — particularly the loss of acetylcholine-producing neurons — and to reduce toxic overstimulation of brain cells by the excitatory neurotransmitter glutamate.
These are FDA-approved and widely prescribed. But their clinical impact is genuinely modest. They do not stop or reverse the disease; at best, they temporarily stabilize some symptoms for some patients. What is more concerning is that recent longitudinal data has raised questions about whether long-term use of these medications might actually be associated with worse outcomes compared to no treatment in some patients — possibly because forcing continued neurotransmission in an actively degenerating brain may hasten the exhaustion of surviving neurons. This is an active area of debate, and the data is not conclusive, but it is a conversation worth having with your neurologist.
The "Magic Bullet" That Never Came
The pursuit of a drug that could halt Alzheimer's by blocking amyloid production consumed an enormous amount of scientific and financial resources over the past two decades. Drugs known as BACE1 inhibitors and Gamma-Secretase Inhibitors were designed to prevent amyloid from forming in the first place. Not only did they fail to slow cognitive decline — multiple trials showed they actively made cognition worse and caused serious side effects including skin cancer and liver damage. This represents one of the highest clinical trial failure rates in modern pharmaceutical history.
The Monoclonal Antibodies: A Real but Limited Advance
The most significant development in conventional Alzheimer's treatment in recent years has been the FDA approval of anti-amyloid monoclonal antibodies — specifically **lecanemab **(brand name Leqembi) and **donanemab **(brand name Kisunla). These intravenous drugs successfully do what decades of research aimed to achieve: they clear amyloid plaques from the brain.
In their pivotal Phase III clinical trials, the results were statistically significant. Lecanemab reduced the rate of clinical decline by 27% on a standard cognitive rating scale (CDR-SB) over 18 months, and donanemab showed a 35% slowing of decline on another validated rating instrument (iADRS). Both drugs also reduced downstream biomarkers associated with neurodegeneration, including plasma p-tau217.
That is genuinely meaningful progress — and it should be acknowledged as such. These are the first treatments that demonstrably modify the trajectory of the disease rather than just masking symptoms.
However, a clear-eyed look at the data reveals important limitations that every patient and family deserves to understand:
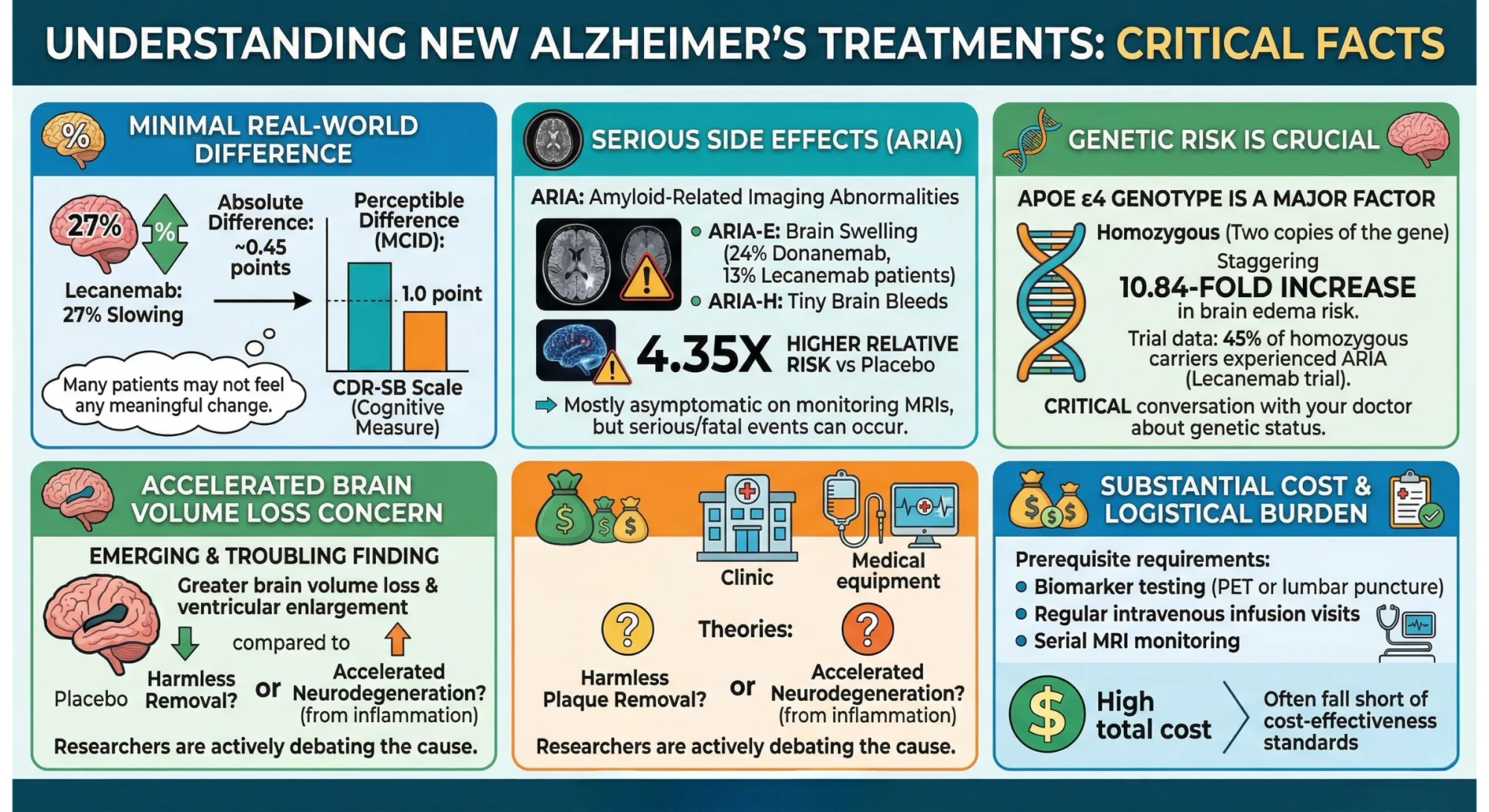
-
The clinical difference is very small in real-world terms. The 27% slowing of decline with lecanemab translated to an absolute difference of approximately 0.45 points on the CDR-SB scale over 18 months. For context, the threshold at which a difference becomes perceptible to patients and families — called the Minimal Clinically Important Difference (MCID) — is 1.0 points. In other words, many patients on this medication would not notice or feel any meaningful change in their daily lives.
-
The side effects are serious. Both drugs cause a condition called Amyloid-Related Imaging Abnormalities (ARIA) — essentially brain swelling (ARIA-E) and tiny brain bleeds (ARIA-H). In clinical trials, ARIA-E occurred in approximately 24% of donanemab patients and 13% of lecanemab patients. Compared to placebo, patients receiving these drugs have a 4.35 times higher relative risk of developing ARIA. Most cases are caught on monitoring MRIs and are asymptomatic, but serious and even fatal events have occurred.
-
Genetic risk matters enormously. Patients who carry the APOE ε4 gene variant — which is also one of the strongest known genetic risk factors for Alzheimer's itself — face dramatically higher risks. Homozygous APOE ε4 carriers (those with two copies of the gene) treated with monoclonal antibodies face a staggering 10.84-fold increased risk of brain edema compared to non-carriers. In the lecanemab trials, 45% of homozygous APOE ε4 carriers experienced ARIA. If you or your loved one has been tested for APOE status, this is a critical conversation to have before starting these medications.
-
Accelerated brain volume loss is a concern. An emerging and troubling finding from both drug trials is that patients receiving anti-amyloid antibodies showed greater-than-expected brain volume loss and ventricular enlargement compared to placebo groups. Researchers are actively debating whether this reflects the harmless physical removal of plaque volume or something more concerning — potentially accelerated neurodegeneration as a consequence of the inflammatory clearance process.
-
The cost and logistical burden are significant. Treatment requires prerequisite biomarker testing (PET scans or lumbar punctures), regular intravenous infusion visits, and serial MRI monitoring. The total cost burden — including the drug, monitoring, and administration — is substantial, and cost-effectiveness analyses have frequently concluded these therapies fall short of standard thresholds.
To summarize: monoclonal antibodies are a genuine scientific achievement that slows disease progression for some patients. But they do not cure Alzheimer's, they do not reverse cognitive decline, and they carry real and serious risks. For many patients, particularly those with APOE ε4 variants or more advanced disease, the risk-benefit calculation is genuinely complicated.
The Functional Medicine Approach: The Bredesen Protocol
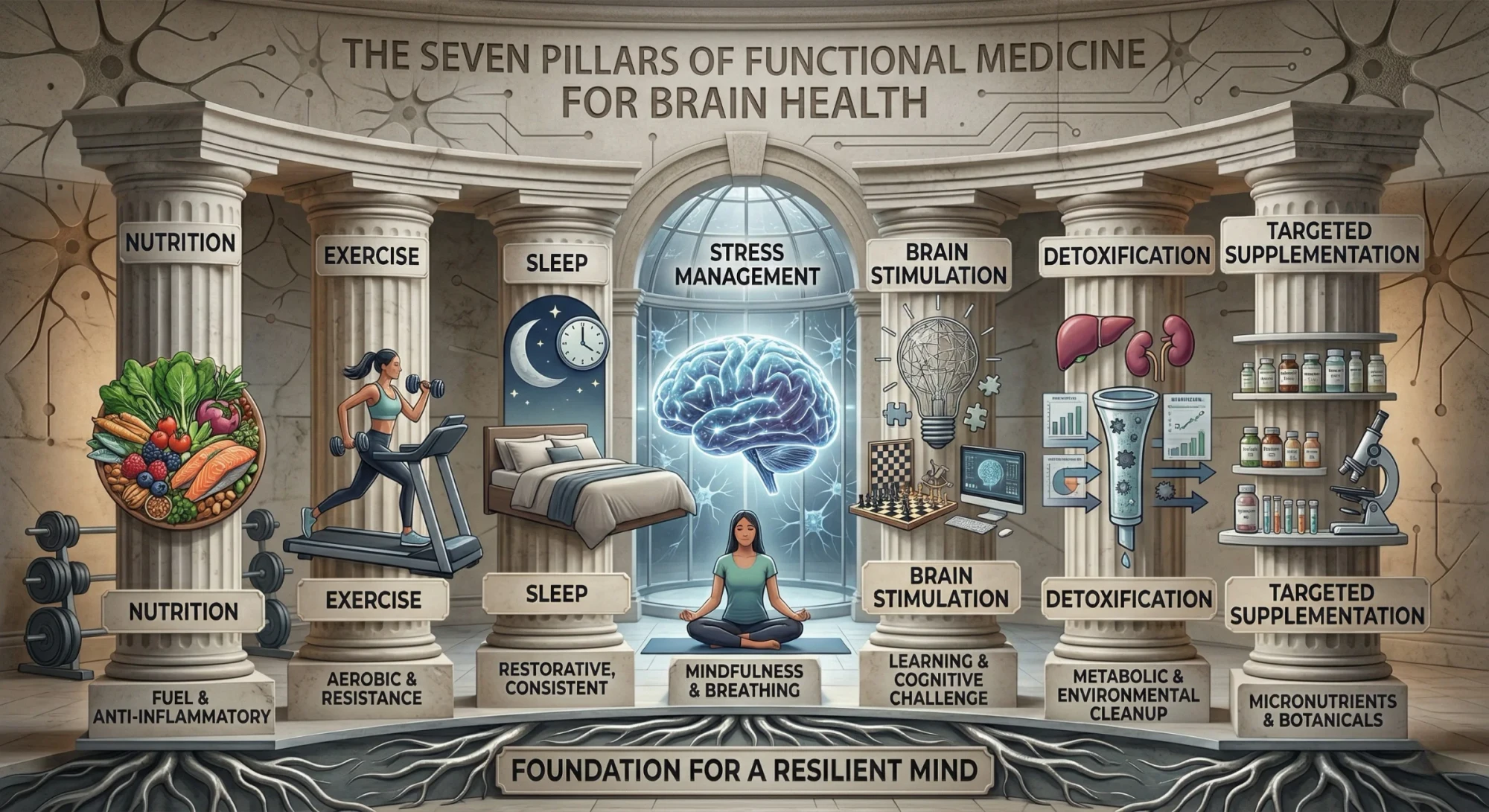
The functional medicine approach to Alzheimer's — most comprehensively developed and studied by Dr. Dale Bredesen — begins from the premise that no single drug can address a disease driven by dozens of intersecting biological failures. Instead of prescribing one intervention, this model prescribes a deeply personalized program targeting the specific combination of metabolic, hormonal, inflammatory, toxic, and lifestyle factors driving neurodegeneration in each individual patient.
The clinical program, known as ReCODE (Reversal of Cognitive Decline) or the Bredesen Protocol, is built around what practitioners call the "Bredesen Seven" — seven core pillars of intervention:
-
Nutrition — typically a ketogenic or Mediterranean-style diet that minimizes blood sugar spikes, reduces inflammation, and provides brain-supportive nutrients.
-
Exercise — regular aerobic and resistance training, both of which have well-documented neuroprotective effects .
-
Sleep Optimization — addressing sleep disorders like sleep apnea and ensuring adequate deep sleep, during which the brain performs critical waste clearance.
-
Stress Management — chronic stress activates inflammatory pathways and disrupts hormonal balance, both of which worsen cognitive function.
-
Brain Stimulation — cognitive engagement, learning, and social connection to maintain neural plasticity.
-
Detoxification — identifying and reducing exposure to environmental toxins that contribute to neuroinflammation.
-
Targeted Supplementation and Hormonal Optimization — correcting specific nutritional deficiencies (Vitamin D, B12, omega-3s, etc.) and addressing hormonal imbalances that are individually identified through comprehensive lab testing.
The key philosophy is personalization. Rather than every patient receiving the same drug, each patient undergoes extensive metabolic and functional testing to identify which of the 36+ known contributors to cognitive decline are active in their particular case. The intervention is then tailored to their unique biology.
What Has the Research Actually Shown?
For many years, the primary evidence supporting the Bredesen Protocol consisted of published case series — collections of individual patient stories showing remarkable cognitive improvements. In 2014, 2016, and 2018, Dr. Bredesen and colleagues published case series detailing improvements in 10 to 100 patients. While these stories were compelling, mainstream neurology — rightly, from a scientific standpoint — was skeptical. Without a control group receiving no treatment, it is genuinely difficult to determine whether improvements were caused by the protocol, by the natural fluctuation of early disease, by the motivating effect of being in a study, or by repeated testing on the same cognitive assessments.
The legitimate scientific gap was the absence of a randomized controlled trial (RCT) — the gold standard for establishing causation in medicine.
The 2025 EVANTHEA Randomized Controlled Trial
In 2025, that gap was addressed. The EVANTHEA trial, led by Toups, Bredesen, and colleagues, was published as the first randomized controlled clinical trial of the precision medicine protocol. While it is important to note this was published as a preprint (meaning it has not yet completed full peer review at time of writing), its findings are scientifically significant and warrant serious attention.
The trial recruited 73 patients aged 45 to 76 with mild cognitive impairment or early-stage Alzheimer's (Montreal Cognitive Assessment scores of 18 or above) across six U.S. clinical sites. Participants were randomized in a 2:1 ratio — 50 patients to the precision medicine protocol and 23 to standard-of-care management. The groups were well-matched for age, education, and genetic risk (46.6% carried at least one APOE ε4 allele).
The results, if they hold up to further scrutiny, are striking:
-
Actual cognitive improvement, not just slower decline. Unlike the monoclonal antibody trials, where the best outcome was a slower rate of worsening, the precision medicine group showed statistically significant cognitive improvement across multiple domains — memory, executive function, processing speed, and overall neurocognitive function — as measured by CNS Vital Signs, a validated digital assessment tool.
-
Meaningful improvements in daily life. Informant-based assessments completed by the patients' partners confirmed significant reductions in perceived disease severity — meaning the improvements were real and noticeable to the people living with these patients.
-
Profound metabolic benefits. The treatment group achieved significant improvements in blood pressure, BMI, fasting glucose, HbA1c (a marker of blood sugar control), homocysteine (a cardiovascular and cognitive risk marker), and vitamin D levels. These are not trivial — metabolic health and brain health are deeply interconnected.
-
Biological aging was reversed. Advanced DNA methylation analysis — a way of estimating a person's biological age based on epigenetic markers — showed that the precision medicine group experienced approximately a 1.3-year reduction in their "biological mortality clock" (OMICmAge). This suggests the protocol did not just slow Alzheimer's progression but may have genuinely reversed some aspects of biological aging.
-
Alzheimer's biomarker reduction. The precision medicine group showed a statistically significant reduction in plasma p-tau217 — the same Alzheimer's-specific biomarker that the monoclonal antibody trials used to demonstrate disease modification. The standard-of-care group did not show this reduction.
-
Effect size comparison. The statistical effect size for overall cognitive function in the precision medicine group was calculated to be approximately seven times greater than that of lecanemab and four times greater than that of donanemab.
-
No serious adverse events. The protocol produced all of these results without a single case of ARIA-E, ARIA-H, or any of the other serious adverse events observed in the drug trials.
Those are remarkable findings. But science demands honest scrutiny, and there are important limitations to acknowledge.
The Debate: Where Critics Have a Point — and Where They Don't
The Bredesen Protocol has faced sustained and sometimes fierce criticism from mainstream neurology, and understanding that criticism is essential for any informed patient.
Legitimate criticisms:
Before the 2025 RCT, the evidence base truly was limited to case series — which, however compelling anecdotally, cannot prove causation. The absence of a control group in earlier publications was a genuine scientific problem, not a minor quibble. The commercialization of the protocol — extensive out-of-pocket costs for testing, supplements, and program subscriptions — has rightly raised concerns about financial accessibility and the potential for exploitation of desperate families. Organizations like the Alzheimer Society of Canada have formally noted that while the lifestyle components of the protocol (diet, exercise, sleep) are well-supported for general health, claims of "reversing" Alzheimer's disease were not validated by traditional standards at the time of their statement.
Regarding the 2025 RCT itself: the sample size of 73 patients is small compared to the thousands enrolled in pharmaceutical trials. The confidence intervals in the cognitive improvement data are wide, meaning there is more uncertainty about the true effect size. The control group's decline may have made the treatment group look more successful by comparison. And the preprint status means the full peer-review process has not yet concluded.
Where the criticism has its own blind spots:
Proponents of the functional medicine approach make a philosophically compelling counter-argument. The randomized, double-blind, placebo-controlled trial — the gold standard in medicine — was designed to test one variable against a single target. It works beautifully for a drug targeting a specific bacteria, for example. But for a complex, multifactorial disease driven by 36 intersecting biological failures, testing one variable at a time is precisely why the pharmaceutical industry has seen a 99% failure rate in Alzheimer's drug development.
Because the precision medicine protocol is personalized — every patient receiving a different combination of interventions based on their unique biology — the data will inherently show more variability than a trial where everyone gets the same pill. This is not a flaw; it is a feature of personalized medicine. The fact that statistically significant, multi-domain cognitive improvements were detectable in a cohort of only 73 patients actually suggests an exceptionally strong signal. By contrast, the monoclonal antibody trials required thousands of patients to demonstrate a difference that most patients cannot feel in their daily lives.

Ultimately, the most honest position a physician can take is this: both paradigms have something real to offer, and both have meaningful limitations. The monoclonal antibodies represent the first drugs that genuinely modify disease biology in a measurable way — but their benefits are modest, their risks are serious, and their costs are prohibitive for many. The functional medicine approach offers compelling evidence of actual cognitive improvement with an excellent safety profile — but the evidence base, while growing, is still not as mature as what is required by traditional pharmaceutical standards. For patients and families navigating this landscape, the goal should not be to choose a camp. The goal should be to work with clinicians who understand both approaches and can help you evaluate which elements make the most sense for your specific situation, genetics, metabolic profile, and values.
What Should You Actually Do? A Practical Framework
Given everything we have covered, here is how I would think about this practically — whether you are a patient, a caregiver, or someone who wants to be proactive about their brain health.
If you have been diagnosed with early Alzheimer's or mild cognitive impairment:
First, ask your neurologist about your APOE genotype if you have not already been tested. This has profound implications for both your disease risk and your risk of serious side effects from monoclonal antibody therapy. If you are an APOE ε4 homozygote, the risk-benefit calculation for these drugs changes dramatically.
Second, recognize that the lifestyle and metabolic pillars of the functional medicine approach are not "alternative" — they are evidence-based. Regular aerobic exercise, a Mediterranean or anti-inflammatory diet, quality sleep, blood sugar control, and management of cardiovascular risk factors all have strong independent evidence for neuroprotection. There is essentially no legitimate scientific argument against implementing these.
Third, if you are interested in the full ReCODE program or a similar precision medicine approach, seek out a clinician trained in functional medicine who can guide comprehensive metabolic testing and personalized intervention. Be informed about the costs, ask for evidence-based explanations of specific recommendations, and be appropriately skeptical of any practitioner who promises certainty.
Fourth, if your neurologist recommends monoclonal antibody therapy, engage seriously with the risk-benefit discussion — particularly around ARIA risk, your APOE status, the need for regular MRI monitoring, and what constitutes meaningful clinical benefit for you personally.
If you are a family member or caregiver:
Your role in this process is invaluable. The informant-based assessments used in research — where partners and family members report changes in the patient's daily function — have consistently proven to be sensitive and meaningful measures. Your observations matter. Advocate for comprehensive evaluation, ask hard questions of the medical team, and do not accept a prescription as the end of the conversation.
If you are healthy but concerned about your future brain health:
This is where the functional medicine framework has perhaps its strongest and least controversial application. The risk factors that drive Alzheimer's — chronic inflammation, poor metabolic health, sleep deprivation, physical inactivity, social isolation, toxin exposure, nutritional deficiencies — are largely modifiable. The 2024 Lancet Commission report identified 14 modifiable risk factors responsible for approximately 45% of Alzheimer's cases globally. Prevention, not treatment, remains the most powerful tool we have.
The Bottom Line
Alzheimer's disease sits at a genuine crossroads in medicine. The standard-of-care approach has, for the first time, produced drugs that modify the disease process — but their benefits remain modest, their risks are significant, and they do not reverse what has already been lost. The functional medicine paradigm offers a compelling alternative framework — one supported by growing evidence, including a landmark 2025 randomized controlled trial — that addresses the disease's underlying metabolic complexity rather than targeting a single biomarker. Its evidence base is still maturing, its costs and logistical demands are real, and the medical establishment's skepticism, while sometimes overstated, is not entirely without merit.
What is clear is this: the search for a single magic bullet to cure Alzheimer's has failed spectacularly and at enormous cost. The most scientifically honest and clinically promising path forward almost certainly involves addressing the full biological, metabolic, and lifestyle landscape of each individual patient — not just the amyloid in their brain. Whether that happens through the evolution of conventional medicine, the growing acceptance of functional medicine, or an integration of both, the patients and families living with this disease deserve nothing less.
Dr. Gina Estupinan.
Whether you choose conventional medicine, a functional approach, or a combination of both, the most important step is taking an informed, proactive role in your brain health journey. You are not powerless — and neither is science.

References
- 1.Zhang, J., Zhang, Y., Wang, J., Xia, Y., Zhang, J., & Chen, L. (2024). Recent advances in Alzheimer’s disease: Mechanisms, clinical trials and new drug development strategies. Signal Transduction and Targeted Therapy, 9(211), 1–35. https://doi.org/10.1038/s41392-024-01911-3
- 2.Toups, K., Tanio, C., Hathaway, A., Bergman, N., Burke, K., Haase, D., Cole, S., Aita, S. L., Raji, C., Boyd, A., Frykman, H., Mammel, A., Elhelou, S., Miller, K., Dwaraka, V., Hausman-Cohen, S., Attarha, M., Mohammadi, S., Sanford, K. ... Bredesen, D. E. (2025). Precision medicine treatment of Alzheimer’s disease: Successful randomized controlled trial [Preprint]. Preprints.org. https://doi.org/10.20944/preprints202512.2694.v1
- 3.Alzheimer's Fast-Changing Treatment Landscape and Pipeline: Need-to-Know Insights, accessed March 2, 2026, https://advisory.avalerehealth.com/insights/alzheimers-fast-changing-treatment-landscape-and-pipeline-need-to-know-insights
- 4.Bredesen, D. E., Ross, M. K., & Ross, S. (2024). Sustained cognitive improvement in Alzheimer’s disease patients following a precision medicine protocol: Case series. Biomedicines, 12(8), 1776. https://doi.org/10.3390/biomedicines12081776
- 5.Alkhalifa, A. E., Al Mokhlf, A., Ali, H., Al-Ghraiybah, N. F., & Syropoulou, V. (2025). Anti-amyloid monoclonal antibodies for Alzheimer’s disease: Evidence, ARIA risk, and precision patient selection. Journal of Personalized Medicine, 15(9), 437. https://doi.org/10.3390/jpm15090437
- 6.Indirect Treatment Comparison of ARIA Outcomes for Lecanemab Compared to Donanemab Based on Reported Results - Eisai Medical, accessed March 2, 2026, https://www.eisaimedical.com/-/media/Files/EisaiMedicalInformation/Neurology/Congress-Materials/AAIC-2025/25-07-24-Dr-Burke-ARIA-ITC---AAIC-Podium_revised.pdf?hash=24308ff6-81fc-4933-ba42-8b33cd8085ba
- 7.Espay, A. J., Kepp, K. P., & Herrup, K. (2024). Lecanemab and donanemab as therapies for Alzheimer's disease: An illustrated perspective on the data. eNeuro, 11(7), ENEURO.0319-23.2024. https://doi.org/10.1523/ENEURO.0319-23.2024
- 8.Re-evaluation of the efficacy and safety of anti-Aβ monoclonal antibodies (lecanemab/donanemab) in the treatment of early Alzheimer's disease - PMC, accessed March 2, 2026, https://pmc.ncbi.nlm.nih.gov/articles/PMC12040886/
- 9.TRAILBLAZER‐ALZ 4: A phase 3 trial comparing donanemab with aducanumab on amyloid plaque clearance in early, symptomatic Alzheimer's disease - PMC, accessed March 2, 2026, https://pmc.ncbi.nlm.nih.gov/articles/PMC12089073/
- 10.Sabbagh, M. N., Cohen, S., Holub, A., Betts, K. A., Rui, S., van Dyck, C. H., & Burke, A. (2025). Indirect treatment comparison of ARIA outcomes for lecanemab compared to donanemab based on reported results. Alzheimer's & Dementia, 21(Suppl 6), e103048. https://doi.org/10.1002/alz70860_103048
- 11.Cummings, J. L., Zhou, Y., Lee, G., Zhong, K., Fonseca, J., Leisgang‐Osse, A. M., & Cheng, F. (2025). Alzheimer's disease drug development pipeline: 2025. Alzheimer's & Dementia: Translational Research & Clinical Interventions, 11(2), Article e70098. https://doi.org/10.1002/trc2.70098
- 12.Rao, R. V., Kumar, S., Gregory, J., Coward, C., Okada, S., Lipa, W., Kelly, L., & Bredesen, D. E. (2021). ReCODE: A personalized, targeted, multi-factorial therapeutic program for reversal of cognitive decline. Biomedicines, 9(10), Article 1348. https://doi.org/10.3390/biomedicines9101348
- 13.Bereczki, E., Mangialasche, F., Barbera, M., Padilla, P., Hara, Y., Fillit, H., Solomon, A., & Kivipelto, M. (2025). Risk reduction and precision prevention across the Alzheimer’s disease continuum: A systematic review of clinical trials combining multidomain lifestyle interventions and pharmacological or nutraceutical approaches. The Journal of Prevention of Alzheimer's Disease, 12(10), Article 100367. https://doi.org/10.1016/j.tjpad.2025.100367
- 14.Mostert, C. M., Udeh-Momoh, C., Winkler, A. S., McLaughlin, C., Eyre, H., Salama, M., Ranchod, K., Trepel, D., Vradenburg, G., Hynes, W., Fieggen, G., Ali, S., EL Mekkaoui, N., Landay, A., Bobrow, K., Muyela, L., Atkins, K., Santuccione Chadha, A., Marongiu, R., . . . Ibanez, A. (2025). Broadening dementia risk models: Building on the 2024 Lancet Commission report for a more inclusive global framework. eBioMedicine, 120, Article 105950. https://doi.org/10.1016/j.ebiom.2025.105950
- 15.Shetty, P., & Youngberg, W. (2018). Clinical lifestyle medicine strategies for preventing and reversing memory loss in Alzheimer’s. American Journal of Lifestyle Medicine, 12(5), 391–395. https://doi.org/10.1177/1559827618766468
