Your Biological Clock: A Secret Superpower You Can Use in Your Favor
What the latest science says about circadian rhythms, meal timing, sleep regularity, and your long-term health
Your body runs on a precise internal clock that governs everything from hormone release to gut health — and modern science shows that aligning your lifestyle with it may be one of the most powerful things you can do for your health.

You have a superpower built right into your biology — and chances are, you have never been formally introduced to it. Every cell in your body keeps time. Not metaphorically, but literally: a molecular clock ticks inside your liver, your gut, your muscles, and your brain, coordinating thousands of biological processes with remarkable precision. Scientists call this your circadian clock, and a rapidly growing body of research suggests that how well you align your daily habits with it may be one of the most consequential decisions you make for your long-term health.
Whether your goal is to perform better athletically, lose weight, reduce your risk of chronic disease, or simply feel more energized and resilient, understanding your circadian biology is not optional — it is foundational. And the good news? The levers you need to pull are not exotic or expensive. They are your meal timing, your sleep schedule, your light exposure, and your movement habits.
What Is the Circadian Clock — and How Does It Work?
At its core, your circadian clock is a network of genes and proteins that create a self-sustaining, roughly 24-hour rhythm inside your cells. The central conductor lives in a tiny region of the brain called the suprachiasmatic nucleus (SCN), located in the hypothalamus. Think of the SCN as the master conductor of an orchestra — it sets the tempo and keeps every musician (your organ clocks) playing in time.
At the molecular level, the clock operates through what scientists call transcription-translation feedback loops. Two proteins — CLOCK and BMAL1 — form a partnership (called a heterodimer) that switches on a set of genes, most notably the Period (Per) and Cryptochrome (Cry) gene families. Once the proteins produced by Per and Cry accumulate to a sufficient level, they team up to switch off the very CLOCK:BMAL1 complex that created them. This elegant on-off cycle takes approximately 24 hours to complete and drives rhythmic changes in virtually every aspect of your physiology.
Critically, these clocks are not isolated from the outside world. They are constantly being reset and synchronized by what scientists call zeitgebers — a German word meaning "time givers." The most powerful zeitgebers for humans are light (which primarily synchronizes the master SCN clock) and food timing (which is especially powerful for synchronizing the peripheral clocks in your liver, gut, muscle, and other organs). When your zeitgebers are consistent and well-timed, your internal clocks hum along in harmony. When they are not — think late-night eating, erratic sleep, or constant exposure to artificial light after dark — your clocks fall out of sync with each other and with the external environment. Scientists call this circadian misalignment, and as we will see, the health consequences are significant.
According to a 2023 review by Sato and Sato published in Endocrinology, the CLOCK:BMAL1 complex directly influences the promoter regions of 10% to 15% of all human genes — meaning roughly one in ten of your genes is under direct circadian control. This is not a minor side process. It is central to how your body regulates metabolism, immunity, hormone release, and cellular repair.
The Key Players: Hormones, Gut, and Muscle All Keep Time
What makes the circadian system so fascinating — and so relevant to your health — is that it does not just govern your sleep. It orchestrates an entire symphony of biological processes that unfold at predictable times of day. Let us look at some of the most important players.
Insulin and GLP-1: Your Metabolic Rhythm Section
Your body processes the same meal very differently depending on what time you eat it. This is not a myth — it is well-established metabolic science. Two hormones are particularly important here: insulin and glucagon-like peptide-1 (GLP-1).
Insulin, produced by the pancreas, is responsible for moving glucose from your blood into your cells for energy or storage. Its effectiveness — what scientists call insulin sensitivity — is not constant throughout the day. It peaks in the morning and declines as evening approaches. GLP-1, a hormone secreted by cells in your intestinal lining, plays a supporting role: it stimulates insulin release in response to food, reduces appetite, and slows gastric emptying. Critically, the beta cells in your pancreas that respond to circulating GLP-1 do so with the greatest sensitivity during the morning hours — a sensitivity that diminishes significantly by evening (Sato & Sato, 2023; BaHammam, 2023).
What this means in practice is that eating the same meal in the morning produces a lower blood sugar spike, a faster and more robust insulin response, and better overall metabolic control compared to eating it at night. Multiple randomized controlled trials reviewed by BaHammam (2023) in Clocks & Sleep confirmed this pattern consistently: identical meals consumed in the evening lead to a considerably greater glycemic response than those consumed in the morning.
Ghrelin, Leptin, and Adiponectin: Your Appetite and Fat Hormones
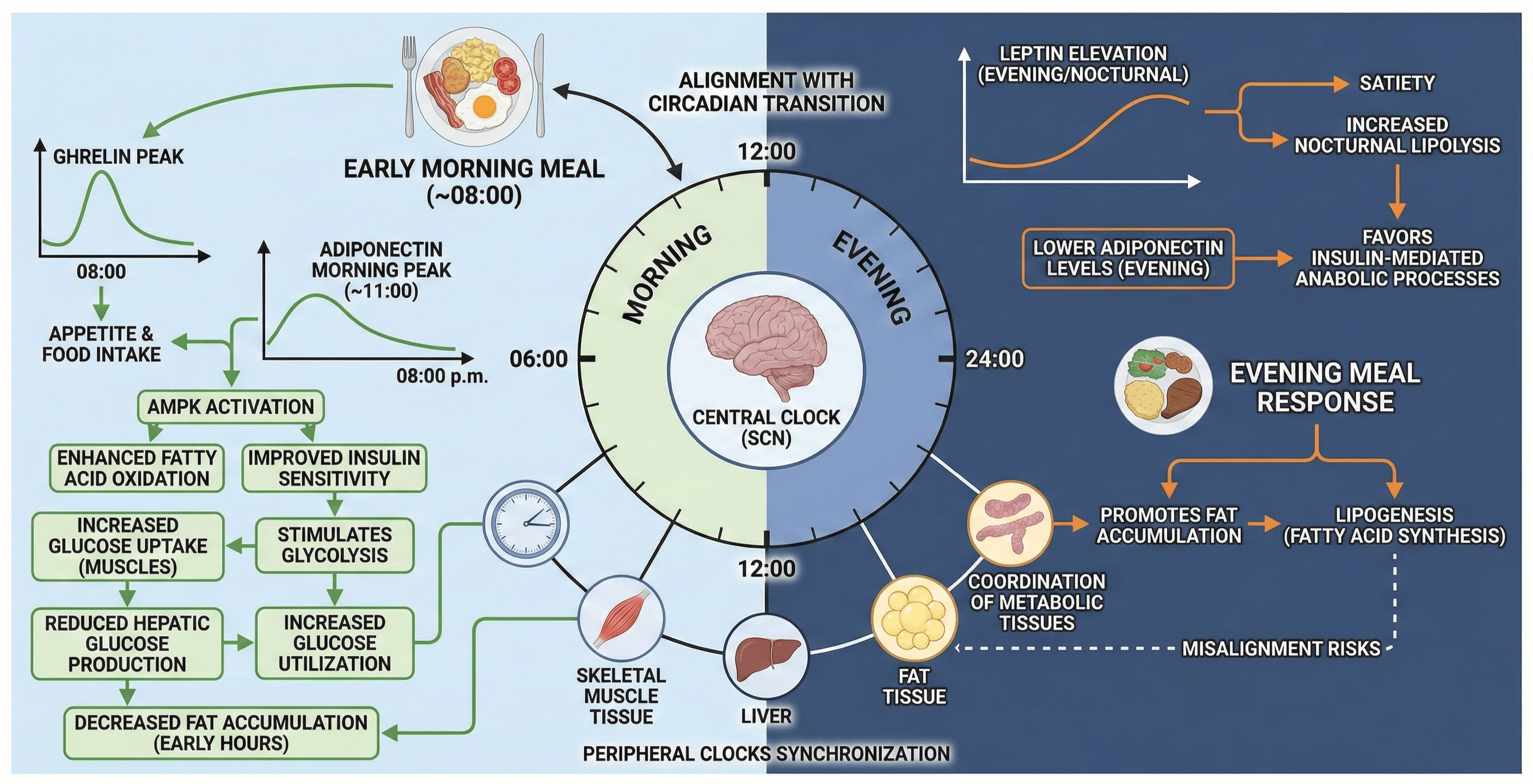
Your hunger hormones also follow a precise circadian schedule. Ghrelin, often called the "hunger hormone," peaks around 8:00 a.m., priming you to eat and helping set feeding-fasting cycles. It stimulates growth hormone release and plays a role in glucose and lipid metabolism — and its rhythmic expression depends on functional clock genes Per1 and Per2. When circadian rhythms are disrupted, ghrelin's pattern flattens, which may contribute to unregulated appetite and metabolic dysfunction.
Adiponectin, a hormone that improves insulin sensitivity and promotes fat oxidation, peaks around 11:00 a.m. before declining by evening. Its morning surge activates AMPK (AMP-activated protein kinase — an enzyme that acts as a cellular energy sensor), enhances fatty acid oxidation, and reduces hepatic glucose production. Conversely, leptin — the satiety hormone — rises in the evening, playing a different metabolic role focused on nocturnal lipolysis (fat breakdown during sleep). Each of these hormones is precisely timed, and late eating disrupts this intricate choreography.
The Gut Microbiome: Your Internal Circadian Ecosystem
Perhaps one of the most surprising frontiers in circadian biology is the gut microbiome — the 10 to 100 trillion microorganisms that live in your gastrointestinal tract. Far from being passive residents, these microbes have their own daily rhythms, and they communicate directly with your circadian clocks.
Your gut bacteria produce metabolites called short-chain fatty acids (SCFAs) — specifically acetate, propionate, and butyrate — through fermentation of dietary fiber. These SCFAs do far more than just nourish your intestinal lining. Research cited by Sato and Sato (2023) demonstrates that SCFAs can actually entrain (synchronize) peripheral clocks in the liver, kidney, and other organs, partly by mediating insulin and GLP-1 secretion. They also influence fat metabolism by acting as selective modulators of PPARγ (a receptor protein involved in fat storage regulation), effectively helping switch your body from fat storage to fat burning.
Critically, the rhythm of microbial activity is highly sensitive to when you eat, not just what you eat. Research shows that feeding-fasting cycles create cyclical changes in gut microflora composition — and disrupting those cycles with erratic eating patterns disrupts the microbial rhythms as well. In animal models, time-restricted feeding (TRF) — limiting food intake to a defined window during the active phase — was shown to decrease the relative abundance of obesity-promoting bacteria while increasing the abundance of obesity-protective strains (Sato & Sato, 2023).

The implications for GI health extend well beyond metabolism. A 2025 review by Bishehsari and colleagues in Gastroenterology drew clear connections between circadian disruption and a range of gastrointestinal conditions. People with inflammatory bowel disease (IBD) show decreased clock gene expression in gut biopsies, and circadian misalignment worsens the chronic inflammatory cycle by reducing SCFA production and impairing the intestinal barrier. Night shift workers face higher rates of erosive esophagitis and GERD, likely due to clock-driven changes in esophageal mucosal integrity. Even colorectal cancer risk appears to be elevated by circadian disruption — late eating (within 3 hours of sleep) was associated with increased risk of colon polyps in human studies, while circadian misalignment promoted tumor progression in animal models.
The gut microbiome is increasingly understood as an active participant in your circadian biology, not a passive bystander. Its rhythmic metabolite production — tightly linked to your feeding schedule — sends signals that help synchronize your liver, your immune system, and your metabolic organs. When your eating window is consistent and aligned with daylight hours, your microbial ecosystem thrives on predictability. When it is not, the downstream consequences touch nearly every corner of your health, from blood sugar regulation to gut inflammation and beyond.
When Clocks Fall Out of Sync: The Health Cost of Circadian Misalignment
The evidence linking circadian disruption to chronic disease has moved well beyond association — it is mechanistic, consistent, and concerning. According to Sato and Sato (2023), an estimated 20% to 25% of adults worldwide already have metabolic syndrome — a cluster of conditions including high blood sugar, excess abdominal fat, elevated blood pressure, and abnormal cholesterol — and modern lifestyle patterns are a major driver.
Shift workers are the most studied population in this area, and the data is sobering. Shift work disrupts melatonin production (melatonin, synthesized from serotonin by the pineal gland, plays a documented role in improving insulin resistance), impairs sleep quality, and chronically alters hormonal rhythms. The meta-analytic evidence shows a 33% higher risk of depressive symptoms in shift workers compared to day workers. Between 15% and 20% of the working population are shift workers — making the public health stakes enormous.
But here is the uncomfortable reality: you do not have to work the night shift to experience circadian disruption. The 2025 review by Bishehsari et al. highlighted that social jetlag — the discrepancy between your weekday and weekend sleep timing — may affect up to 80% of individuals. Scrolling your phone in bed, eating dinner at 9 p.m., sleeping in on Sundays, and having your bedroom lit up by streetlights at night are all forms of mild but cumulative circadian disruption that, over years, add up to meaningful metabolic and health risk.
Sleep Regularity: More Powerful Than You Think
Most conversations about sleep focus on duration — are you getting 7 to 8 hours? But a landmark 2024 study published in Sleep by Windred and colleagues suggests that regularity — going to bed and waking up at consistent times day after day — may actually matter more for long-term survival.
The researchers analyzed over 10 million hours of objective accelerometer data from nearly 61,000 participants in the UK Biobank and tracked mortality over more than 6 years. Their findings were striking. Compared to those with the least consistent sleep schedules, individuals with the most regular sleep patterns showed:
- 20% to 48% lower risk of all-cause mortality - 16% to 39% lower risk of cancer-related death - 22% to 57% lower risk of cardiometabolic death

Perhaps most remarkably, when sleep regularity was accounted for, sleep duration alone did not explain significant additional variance in mortality risk. In other words, a consistent 6.5 hours every night may carry more protective value than a variable schedule that averages 8 hours. This finding reinforces the core principle of circadian biology: your body thrives on predictability and timing consistency, not just volume.
The Science of Meal Timing: When You Eat Is as Important as What You Eat
The convergence of evidence on meal timing is perhaps the most actionable insight from modern circadian science. Several distinct but complementary lines of research now point to the same conclusion: aligning your food intake with the early, active phase of the day is one of the most powerful metabolic interventions available to you.
Early Eating and Glucose Control
A study analyzed in BaHammam's 2023 narrative review directly tested what happens when you skip breakfast versus eating it at 8:00 a.m. The researchers found that extended fasting until noon decreased mRNA expression of key clock genes — including AMPK, BMAL1, PER1, and RORα — before and after lunch. This fasting pattern was linked to elevated post-meal blood sugar levels and impaired, delayed insulin and GLP-1 responses at lunchtime.
In contrast, breaking the overnight fast with a substantive breakfast at 8:00 a.m. had a "resetting" effect on these metabolic clock genes. The result? Lower blood sugar after lunch, faster and more robust insulin secretion, and better GLP-1 responses — all from the same lunch meal, simply by having eaten breakfast earlier. This finding elegantly illustrates how meal timing acts as a zeitgeber for your peripheral metabolic clocks.
Time-Restricted Eating and Cardiometabolic Health
A 2026 randomized controlled trial by Grimaldi and colleagues published in Arteriosclerosis, Thrombosis, and Vascular Biology took a fresh and highly practical approach to time-restricted eating. Rather than imposing a rigid fasting window, they asked participants to simply eat their last meal at least 3 hours before their habitual sleep time, extending the overnight fast to between 13 and 16 hours.
Over 7.5 weeks, compared to a control group, this sleep-aligned approach produced meaningful improvements:
- Lower nighttime heart rate and higher heart rate variability (a marker of healthy autonomic nervous system function) - Improved diastolic blood pressure dipping during sleep (a critical marker of cardiometabolic health) - Lower nighttime cortisol levels - Lower blood glucose and improved acute insulin response during an oral glucose tolerance test the following morning
What makes this study particularly compelling is that the intervention was modest and individualized — not a one-size-fits-all 16-hour fast, but simply a 3-hour buffer before sleep. This is a genuinely accessible lifestyle change that works with your biology rather than against it.
Meal Timing and Genetic Risk: When Timing Can Override Your DNA
One of the most thought-provoking recent findings comes from a 2025 study published in Obesity by De la Peña-Armada and colleagues. This study followed 1,195 adults and calculated each participant's genetic predisposition to obesity using a polygenic risk score (PRS-BMI) — essentially a composite of nearly one million small genetic variations that collectively influence body weight.
The researchers then examined how the midpoint of daily meal intake (the halfway point between first and last meal) interacted with this genetic risk. Their findings revealed a striking gene-environment interaction:
- For individuals with a high polygenic risk for obesity, every hour of delay in meal midpoint was associated with a BMI increase of more than 2 kg/m². But when these high-risk individuals ate early, their BMI was significantly lower — effectively attenuating the expression of their obesity-prone genes. - For individuals with a low genetic risk, meal timing had almost no impact on BMI. Their metabolism was more resilient to late eating.
Looking at 12-year follow-up data from a weight-loss maintenance program, the researchers confirmed that later eating was a significant predictor of long-term weight regain. Each one-hour delay in meal midpoint was associated with a 2.2% higher body weight over time.
This is a profound finding. It tells us that chrononutrition — the timing of meals relative to circadian rhythms — is not equally important for everyone. For those with a genetic tendency toward obesity, when you eat may be just as critical as what you eat. It also opens the door to a more personalized approach: using genetic profiling to identify who would benefit most from a strict early-eating protocol.
Practical Lifestyle Interventions: What the Evidence Recommends
Let us synthesize the key takeaways from this body of research into concrete, actionable strategies. None of these require a prescription or a laboratory — only intention and consistency.
1. Eat earlier in the day, and keep your eating window consistent. Aim to have your first meal relatively early (around 7:00–9:00 a.m.) and your last meal at least 3 hours before your typical bedtime. This aligns your food intake with peak insulin sensitivity, optimal GLP-1 response, and the hormonal rhythms your body has evolved to use. The research consistently shows that early time-restricted eating improves glucose tolerance, reduces blood pressure, and supports healthy body weight — particularly for those with metabolic risk factors.
2. Prioritize sleep regularity above all else. Choose a consistent bedtime and wake time and defend them — even on weekends. The UK Biobank data is unambiguous: the regularity of your sleep schedule is a more powerful predictor of mortality risk than the number of hours you sleep. If you can only make one sleep change, make it this one.
3. Stop eating at least 3 hours before bed. The Grimaldi (2026) trial demonstrated that this simple change — individualized to your own sleep schedule — improves nighttime autonomic balance, lowers blood pressure, reduces cortisol, and enhances morning glucose tolerance. You do not need a rigid fasting protocol. You simply need to close your eating window well before you sleep.
4. Protect your light environment. Light is the primary zeitgeber for your master clock. Get bright, natural light exposure in the morning (ideally within an hour of waking) to anchor your circadian rhythm. In the evening, dim your lights 2 to 3 hours before bed and minimize exposure to blue-light-emitting screens. This is not aesthetic preference — it directly influences melatonin secretion, sleep quality, and downstream metabolic function.
5. Feed your gut microbiome with timing in mind. Consistent feeding-fasting cycles create predictable microbial rhythms that support SCFA production, gut barrier integrity, and peripheral clock entrainment. A diet rich in fiber — vegetables, legumes, whole grains — provides the substrate your gut bacteria need to produce the SCFAs that support metabolic health. Eating that fiber-rich food during the day, not late at night, amplifies those benefits.
6. Time your exercise thoughtfully. Skeletal muscle releases signaling molecules called myokines — including interleukin-6 (IL-6) and beta-aminoisobutyric acid (BAIBA) — that support fat oxidation, glucose uptake, and metabolic communication between organs. Research shows that both the production of these myokines and their effects on metabolism are time-dependent. While the optimal exercise window likely varies by individual and goal, avoiding late-night high-intensity exercise (which can elevate cortisol and delay sleep onset) is a sensible default.
7. If you are a shift worker or frequent traveler, be proactive. The negative health effects of shift work are real, but they are not inevitable. Strategies such as controlled light exposure during your shift, keeping meal timing as consistent as possible relative to your shifted schedule, and prioritizing sleep regularity even within a non-traditional schedule can meaningfully reduce the metabolic burden of circadian disruption.
The Bottom Line: Your Clock Is Already Working — Help It Help You
Your biological clock is not a passive timekeeper. It is an active, intelligent system that coordinates your hormones, your gut microbiome, your immune function, your cardiovascular system, and your metabolism — all in service of keeping you healthy. The science reviewed here, drawn from multiple rigorous studies published between 2023 and 2026, makes one thing abundantly clear: the timing of your behaviors matters as much as the behaviors themselves.
You do not need to overhaul your entire life overnight. Start with one or two anchoring habits — a consistent wake time, a last meal at least 3 hours before bed, a morning breakfast that resets your metabolic clocks. Over time, these small, consistent shifts compound into genuine biological change. Your circadian clock is already working for you. The question is simply whether you are working with it.
Dr. Gina Estupinan.
Your biology is not working against you — it is waiting for you to work with it. Start small, stay consistent, and let your clock become your greatest ally.

References
- 1.Sato, T., & Sato, S. (2023). Circadian regulation of metabolism: Commitment to health and disease. Endocrinology, 164(7), 1–12. https://doi.org/10.1210/endocr/bqad086
- 2.BaHammam, A. S. (2023). Timing matters: The interplay between early mealtime, circadian rhythms, gene expression, circadian hormones, and metabolism—A narrative review. Clocks & Sleep, 5(3), 507–535. https://doi.org/10.3390/clockssleep5030034
- 3.De la Peña-Armada, R., Rodríguez-Martín, M., Dashti, H. S., Cascales, A. I., Scheer, F. A. J. L., Saxena, R., & Garaulet, M. (2025). Early meal timing attenuates high polygenic risk of obesity. Obesity (Silver Spring), 33(8), 1555–1566. https://doi.org/10.1002/oby.24319
- 4.Grimaldi, D. (2026). Sleep-aligned extended overnight fasting improves nighttime and daytime cardiometabolic function. Arteriosclerosis, Thrombosis, and Vascular Biology. Advance online publication. https://doi.org/10.1161/ATVBAHA.125.323355
- 5.Windred, D. P., Burns, A. C., Lane, J. M., Saxena, R., Rutter, M. K., Cain, S. W., & Phillips, A. J. K. (2024). Sleep regularity is a stronger predictor of mortality risk than sleep duration: A prospective cohort study. Sleep, 47(1), zsad253. https://doi.org/10.1093/sleep/zsad253
- 6.Bishehsari, F., Post, Z., Swanson, G. R., & Keshavarzian, A. (2025). Circadian rhythms in gastroenterology: The biological clock's impact on gut health. Gastroenterology, 169(7), 1380–1396. https://doi.org/10.1053/j.gastro.2025.06.017
